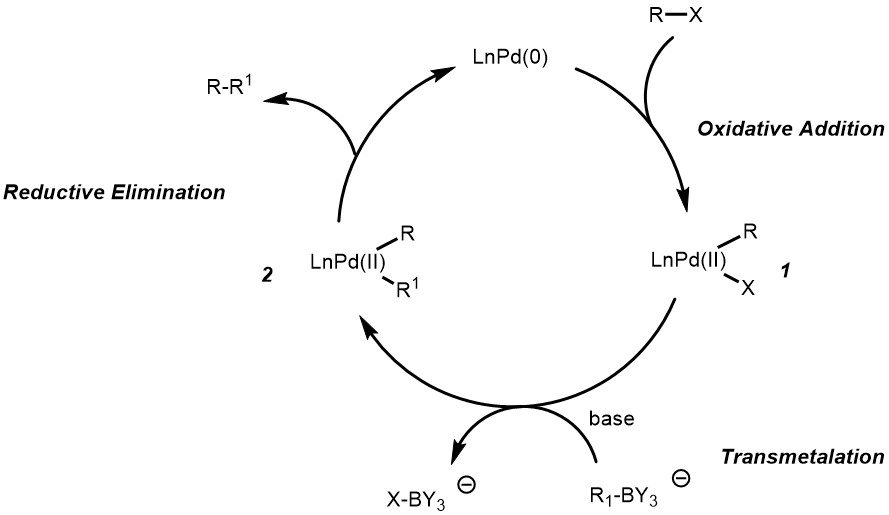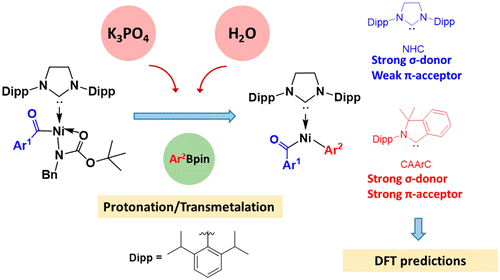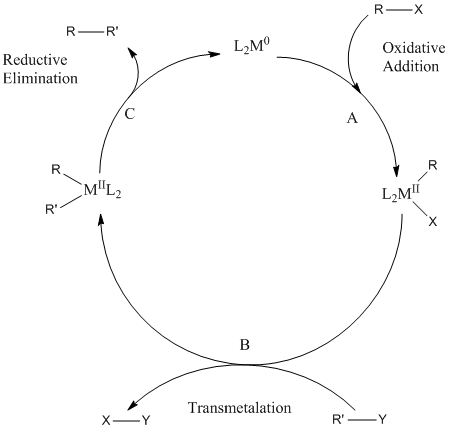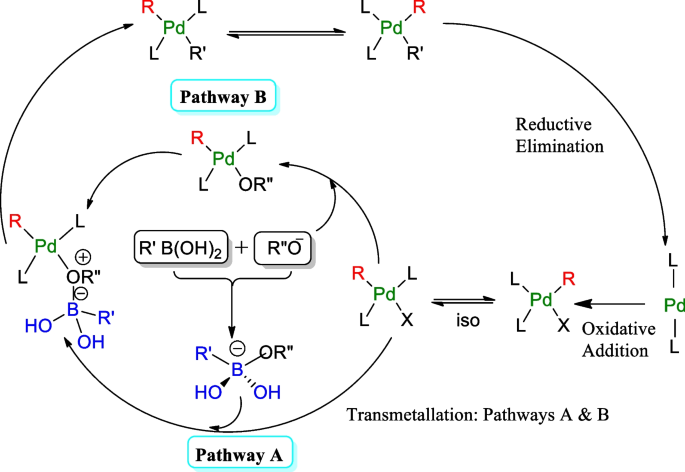
Suzuki–Miyaura cross-couplings for alkyl boron reagent: recent developments—a review | Future Journal of Pharmaceutical Sciences | Full Text
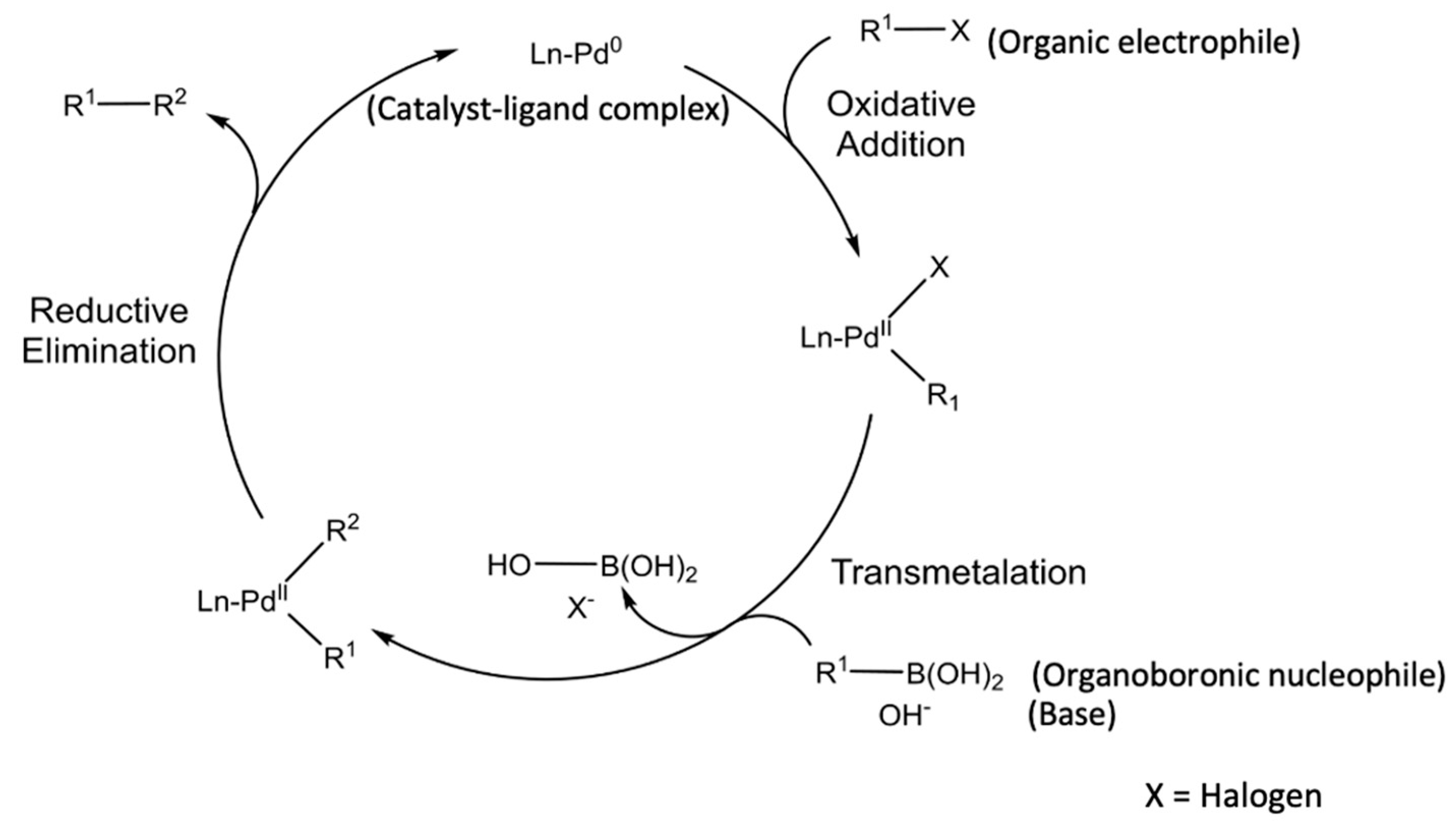
Knowledge | Free Full-Text | Catalyst Recycling in the Suzuki Coupling Reaction: Toward a Greener Synthesis in the Pharmaceutical Industry
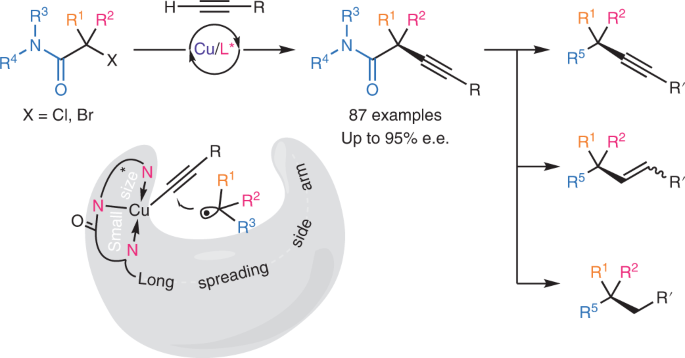
Mechanism-based ligand design for copper-catalysed enantioconvergent C(sp3)–C(sp) cross-coupling of tertiary electrophiles with alkynes | Nature Chemistry

Pd-NHC catalyzed Suzuki cross-coupling of benzyl ammonium salts | Research on Chemical Intermediates
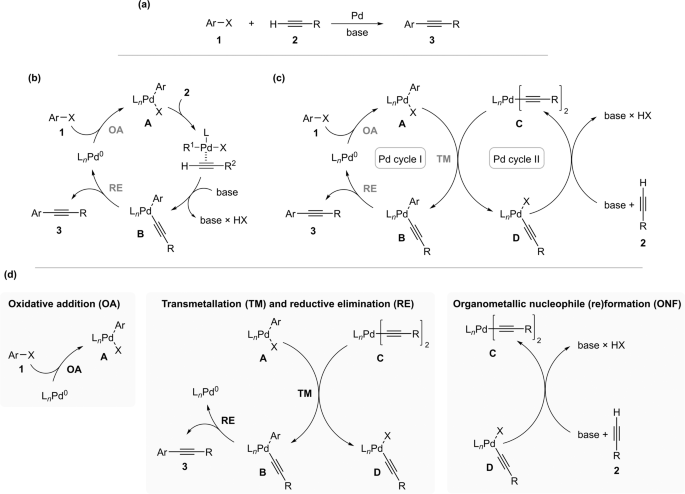
Elucidating the reaction mechanism of a palladium-palladium dual catalytic process through kinetic studies of proposed elementary steps | Communications Chemistry

Molecules | Free Full-Text | Nickel-Catalyzed Suzuki Coupling of Phenols Enabled by SuFEx of Tosyl Fluoride

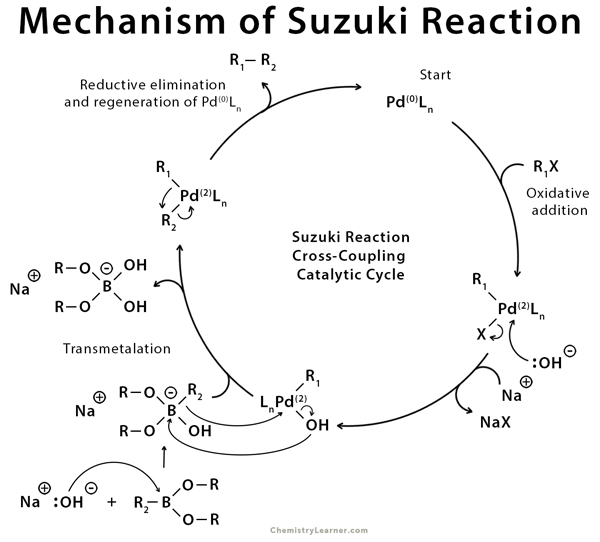
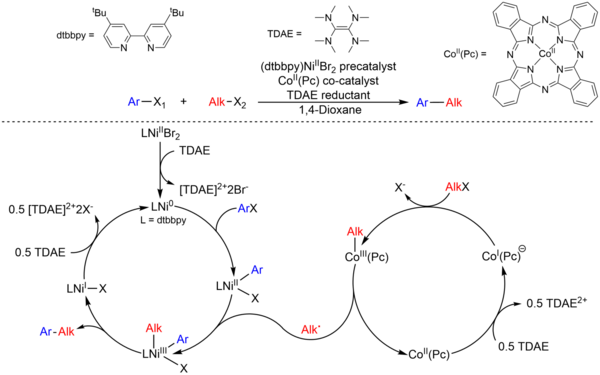
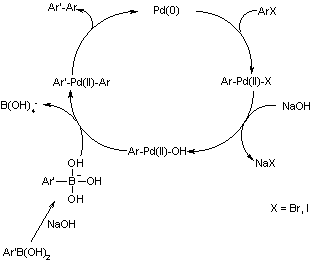
![46] Suzuki Cross Coupling 1979 – ChemInfoGraphic 46] Suzuki Cross Coupling 1979 – ChemInfoGraphic](https://cheminfographic.wordpress.com/wp-content/uploads/2017/11/46_suzuki_coupling1.jpg)