
does mohr's salt contain iron ? write the chemical formula of mohr's salt? from brainly - Brainly.in
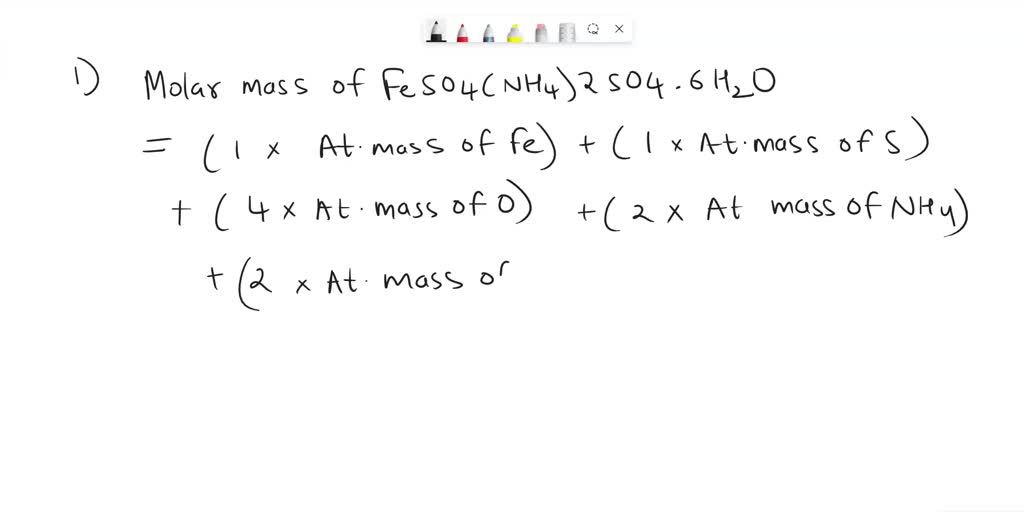
SOLVED: Calculate the percentage of iron in Mohr salt, if the molecular formula is FeSO4-(NH4)2SO4.6H2O. Comment on how this value compares with your experimental value. Draw the Lewis structure of the sulfate

The molecular formula of Mohr's salt is (NH.),SO.FeS0.6H,O(1) Find the number of atoms of each element.(2) - Brainly.in

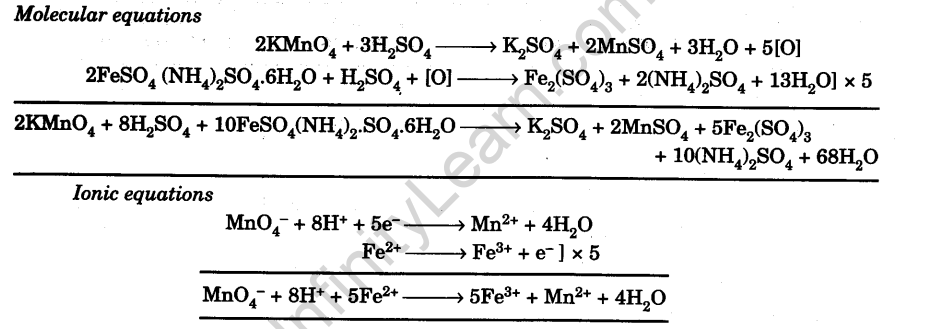
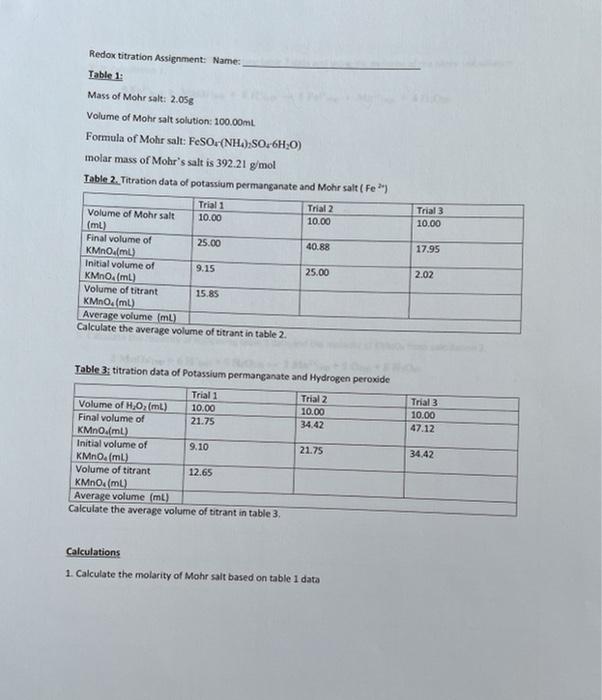





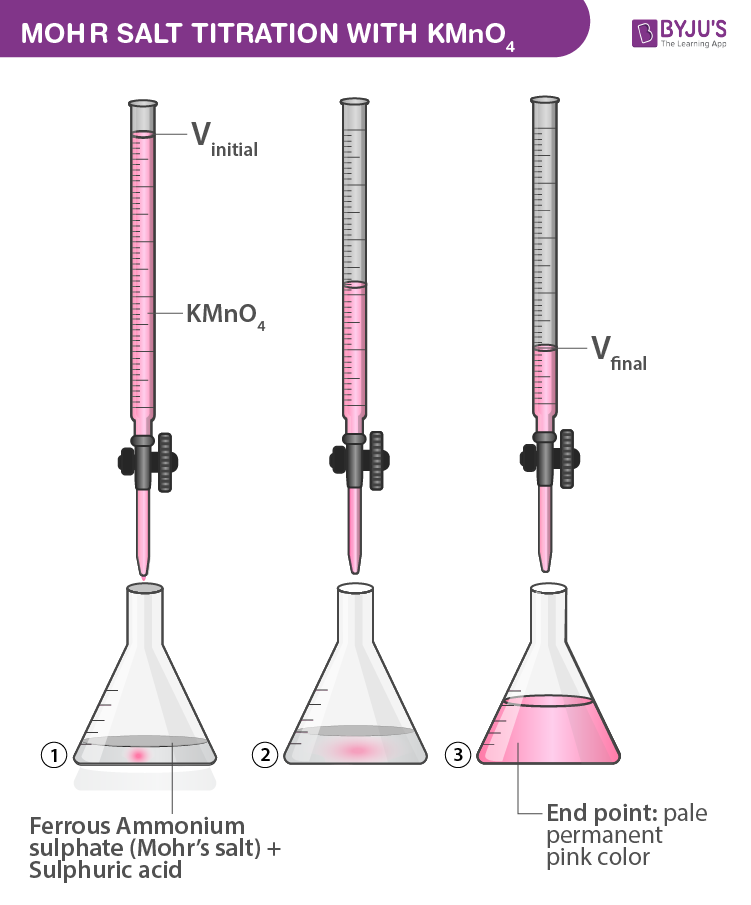









![Odia] What is formula of Mohr's salt. Odia] What is formula of Mohr's salt.](https://static.doubtnut.com/ss/web/11457989.webp)
