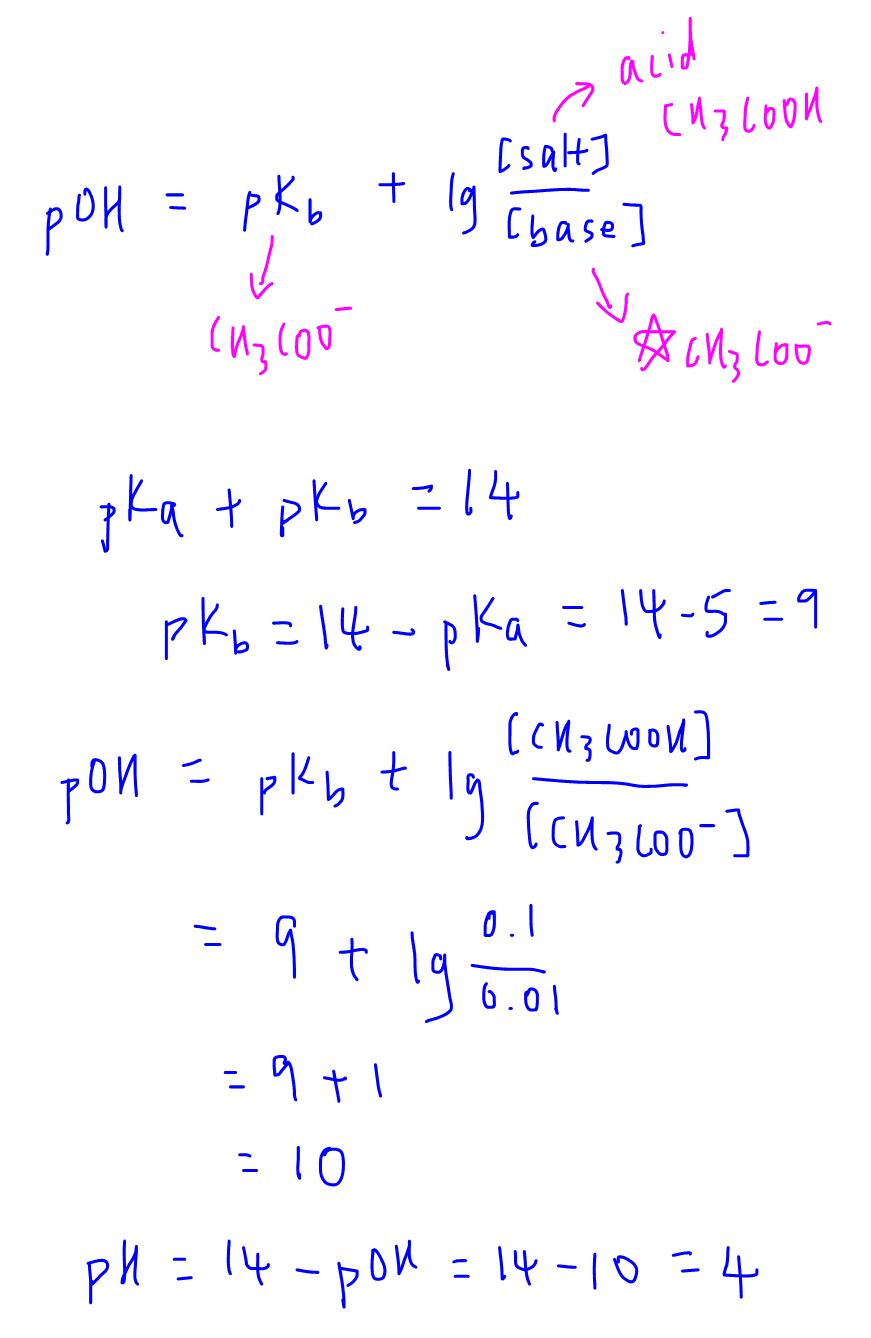equilibrium - Calculation of the pH of a mixture of a strong acid and weak acid - Chemistry Stack Exchange
![PH calculations. What is pH? pH = - log 10 [H + (aq) ] where [H + ] is the concentration of hydrogen ions in mol dm -3 to convert pH into hydrogen ion. - ppt download PH calculations. What is pH? pH = - log 10 [H + (aq) ] where [H + ] is the concentration of hydrogen ions in mol dm -3 to convert pH into hydrogen ion. - ppt download](https://images.slideplayer.com/24/7441095/slides/slide_21.jpg)
PH calculations. What is pH? pH = - log 10 [H + (aq) ] where [H + ] is the concentration of hydrogen ions in mol dm -3 to convert pH into hydrogen ion. - ppt download
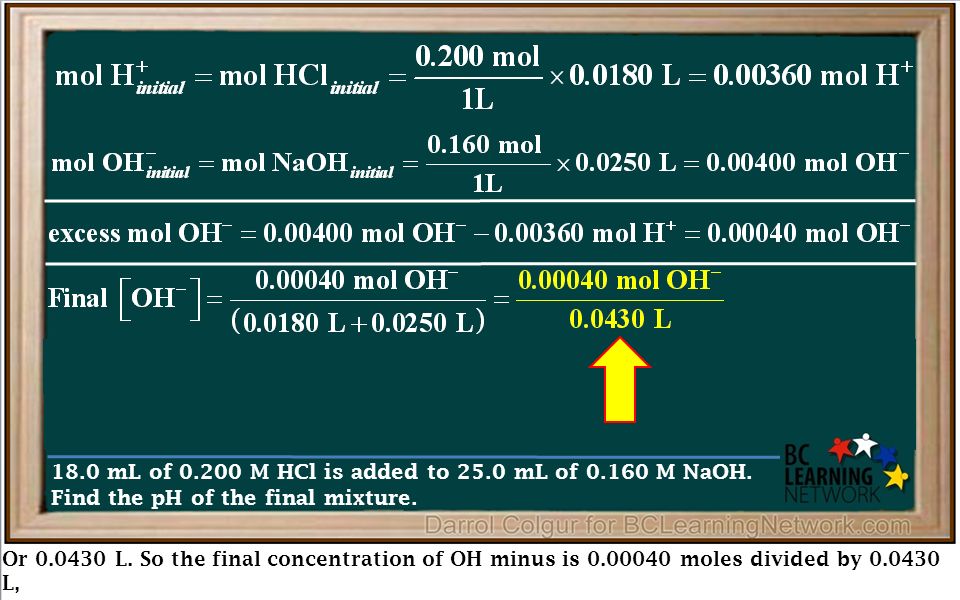
We'll show you how to calculate the pH of a solution formed by mixing a strong acid with a strong base. Strong Acid– Strong Base Mixtures. - ppt download