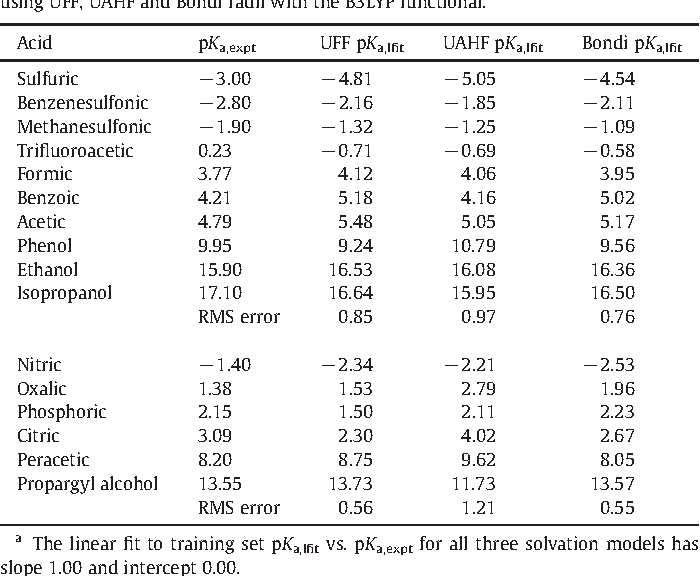
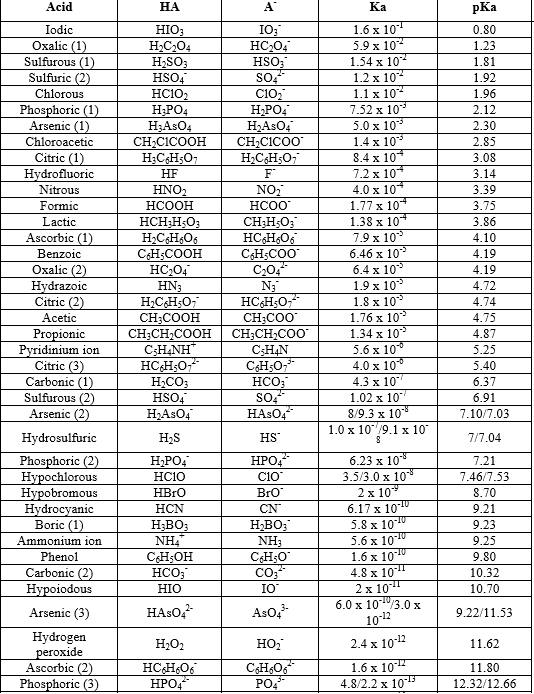
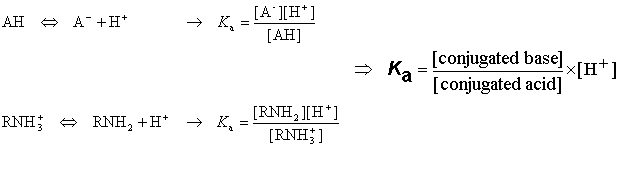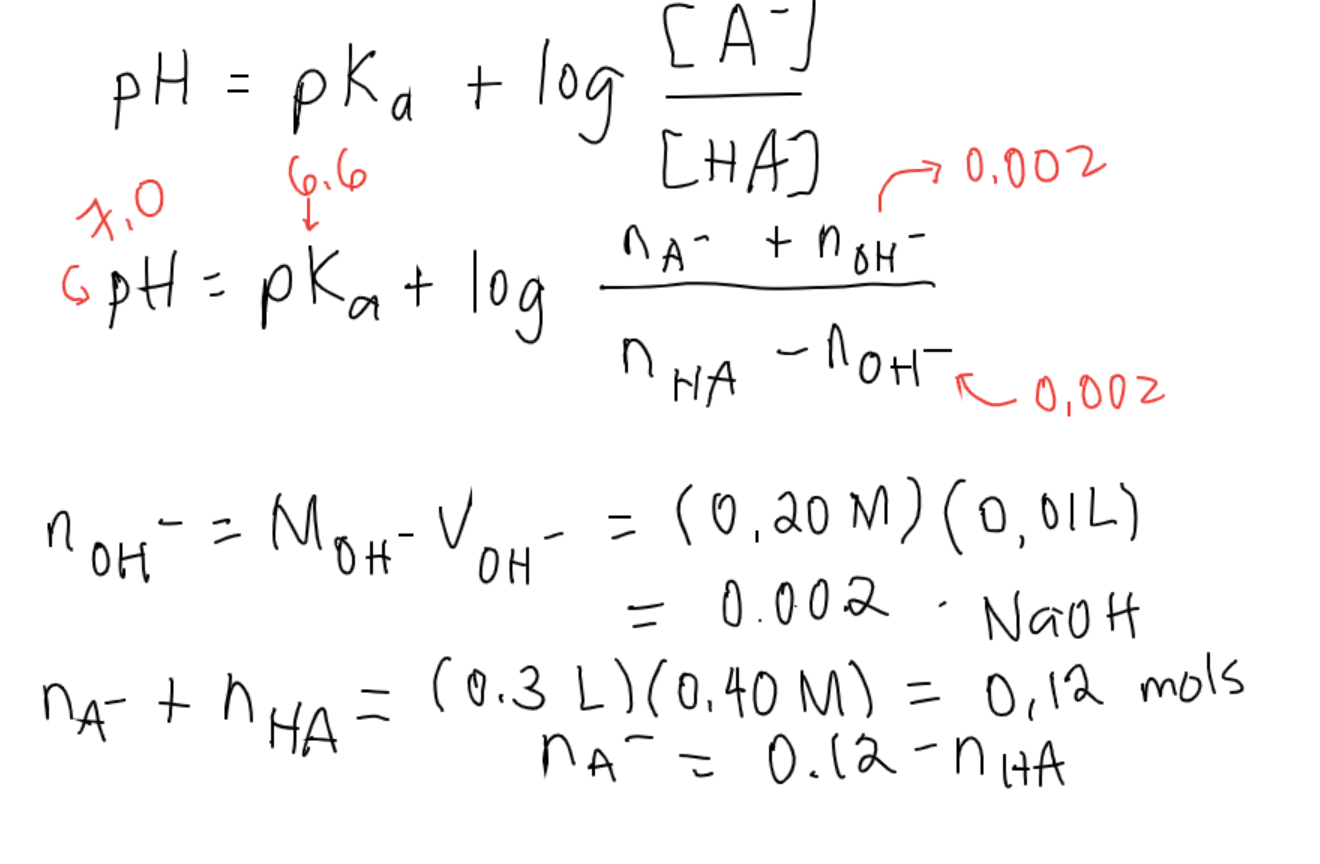On the Accuracy of the Direct Method to Calculate pKa from Electronic Structure Calculations | The Journal of Physical Chemistry A

SOLVED: pH = pKa + log10 Using the Henderson-Hasselbalch equation as seen above, solve for pKa. Given the mixture of 0.30 M sodium acetate and 0.20 M acetic acid, what is the





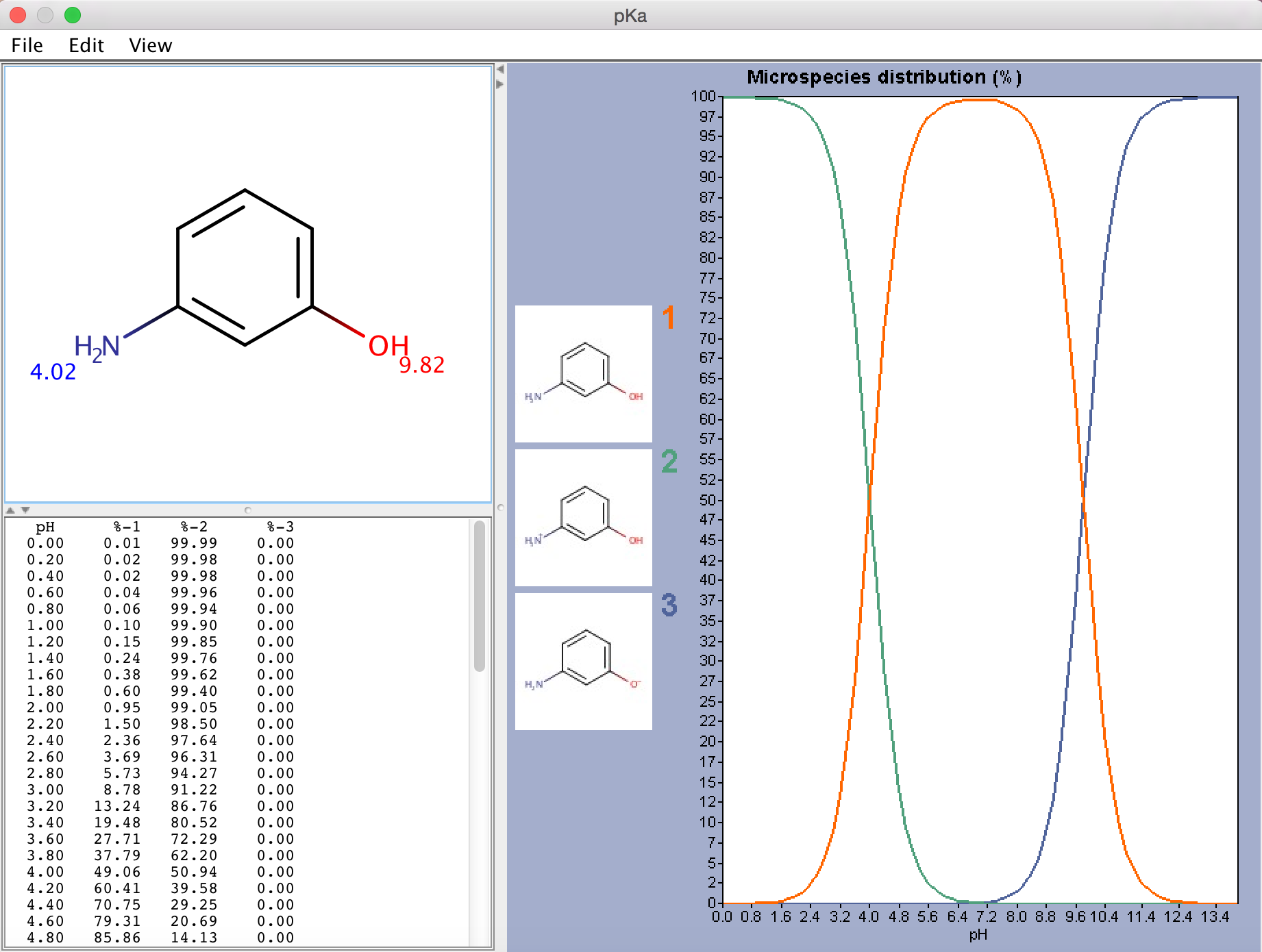
![chemaxoncalcpka [online documentation] chemaxoncalcpka [online documentation]](https://doc.mcule.com/lib/exe/fetch.php?w=800&tok=562d71&media=pka.png)