Photoinduced ring opening and closure reactions in chalcones monitored by time-resolved IR spectroscopy
Transition state models for Baldwin's rules of ring closure. - Henry Rzepa's Blog Henry Rzepa's Blog

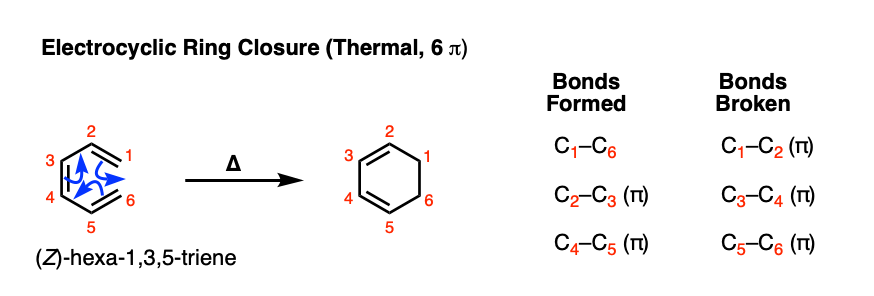
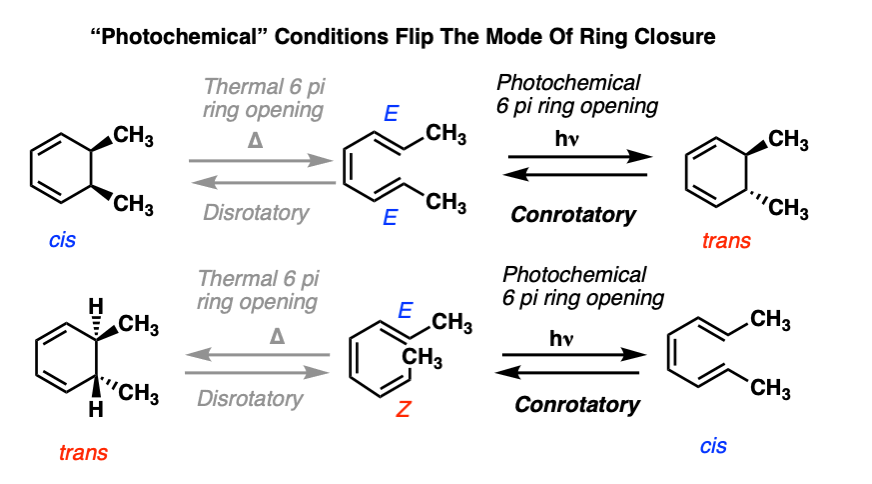
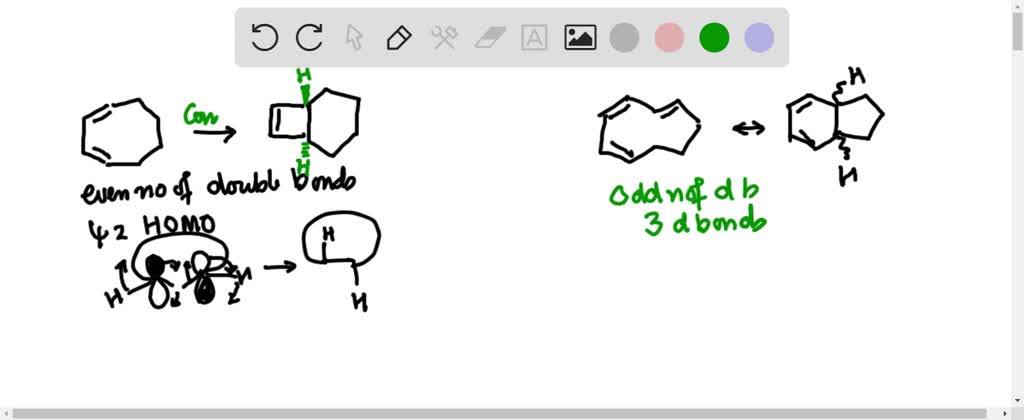
Consider the following electrocyclic ring closure. Does the product form by a conrotatory or disrotatory process? Would this reaction occur under photochemical or thermal conditions? | Homework.Study.com

Mechanism of Oxidative Ring‐Closure as Part of the Hygromycin Biosynthesis Step by a Nonheme Iron Dioxygenase - Ali - 2021 - ChemCatChem - Wiley Online Library
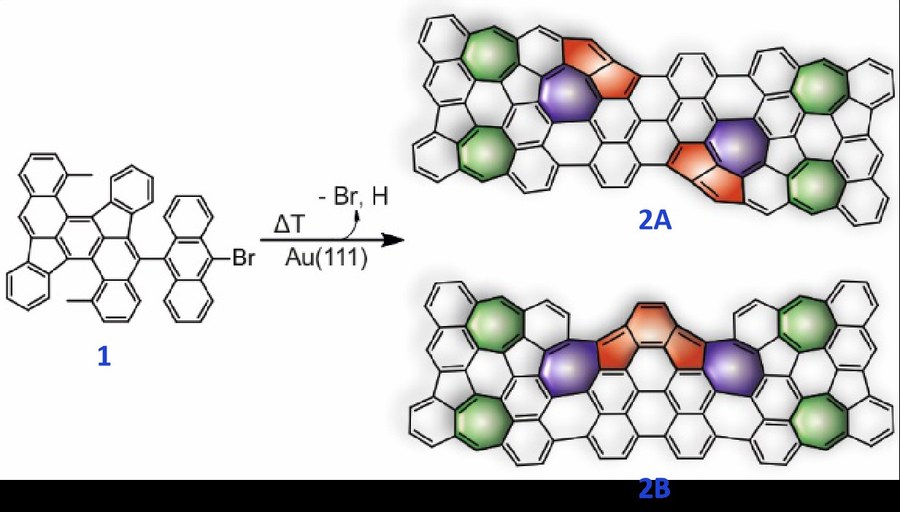
On-Surface Synthesis of Non-Benzenoid Nanographenes by Oxidative Ring- Closure and Ring-Rearrangement Reactions — Professur für Molekulare Funktionsmaterialien — TU Dresden
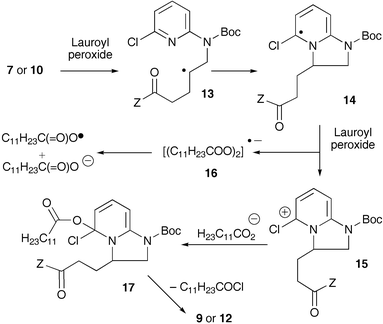
An unprecedented radical ring closure on a pyridine nitrogen - Chemical Communications (RSC Publishing) DOI:10.1039/B609021D

JCM | Free Full-Text | Effective and Secure Closure after Duodenal Endoscopic Submucosal Dissection: Combination of Endoscopic Ligation with O-Ring Closure and Over-the-Scope Clip
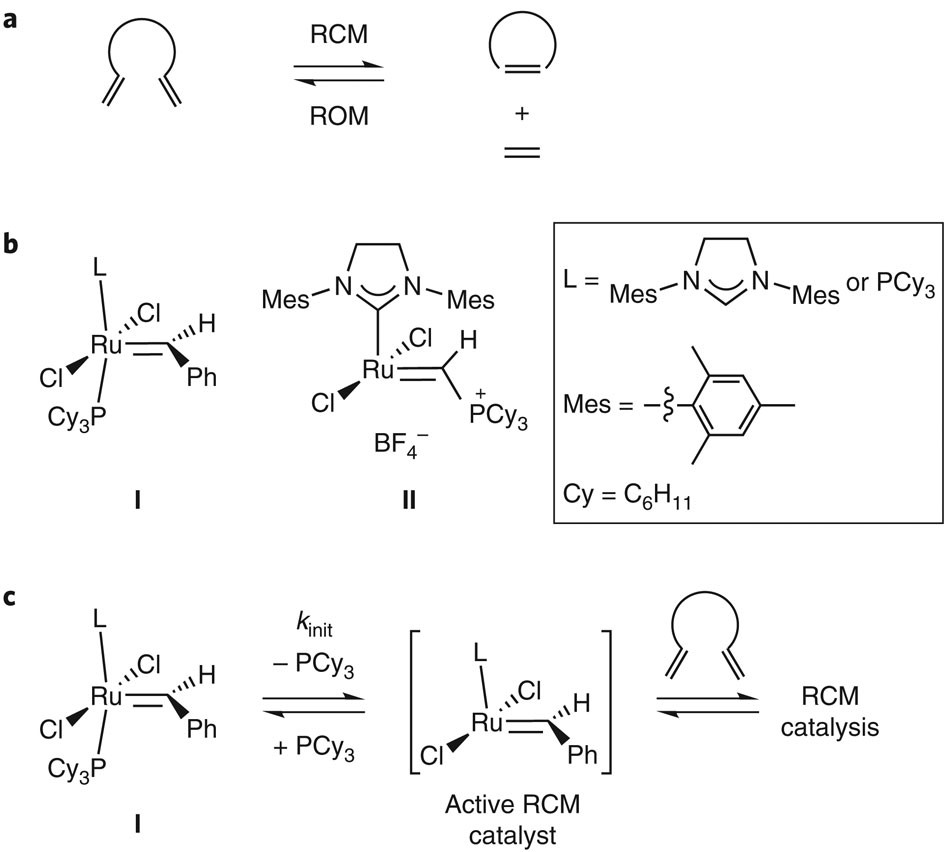
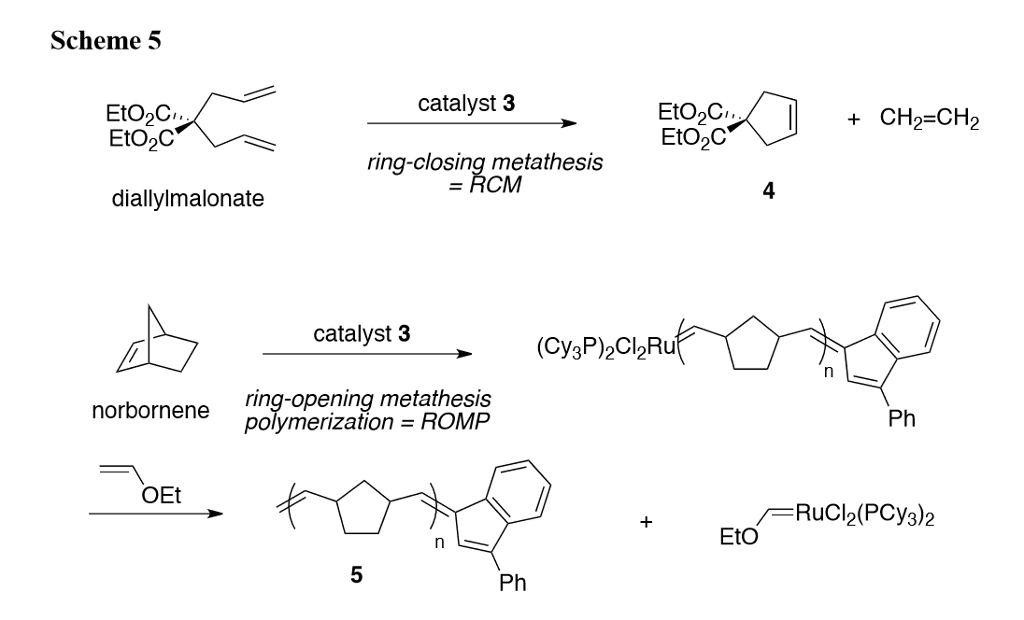
Mechanistic insights into the ruthenium-catalysed diene ring-closing metathesis reaction | Nature Chemistry

Consider the following electrocyclic ring closure. Does the product form by a conrotatory or disrotatory process? Would this reaction occur under photochemical or thermal conditions? | Homework.Study.com