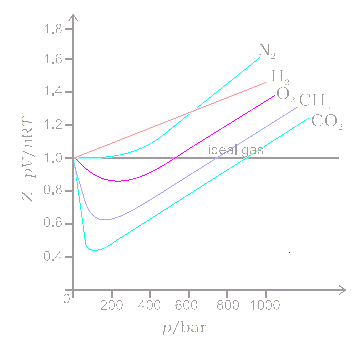
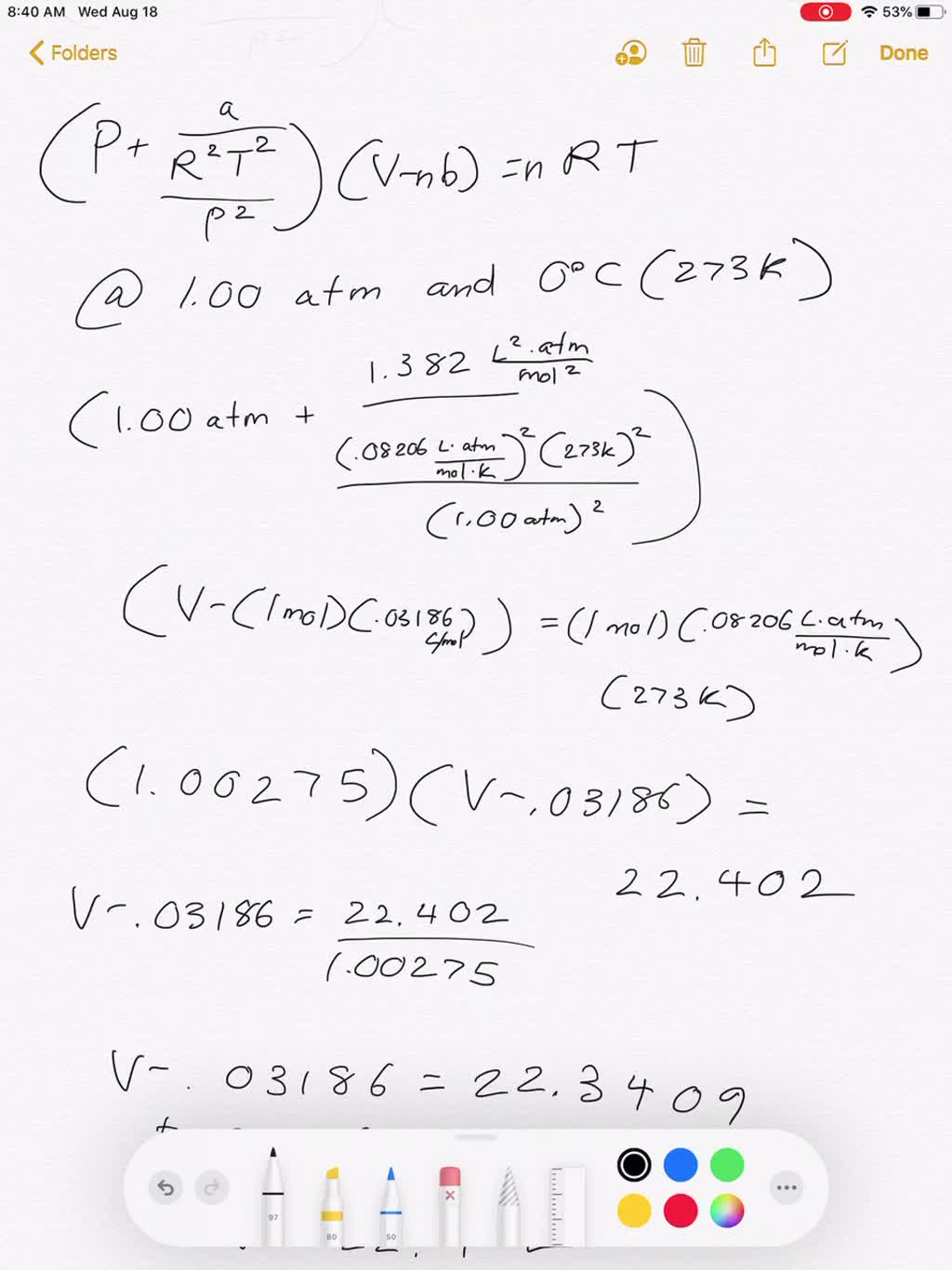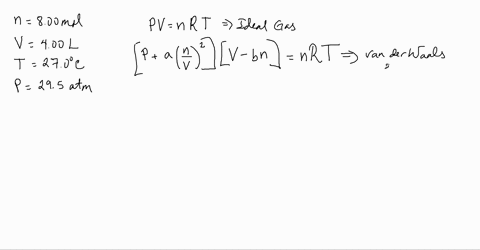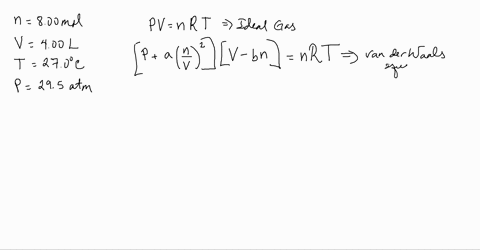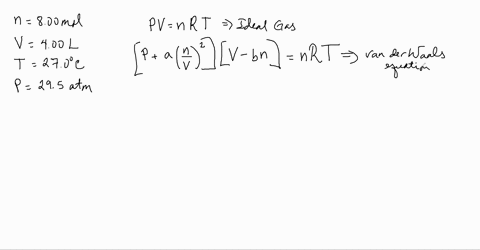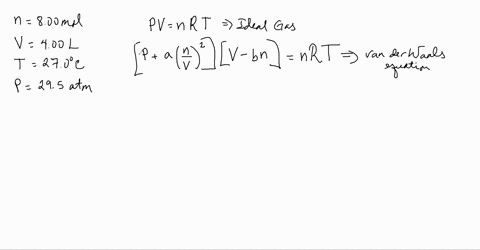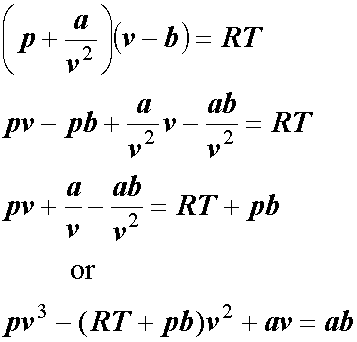3-4. Using the van der Waals equation, calculate the pressure of 10.0 mol NH: gas in a 10.0 L vessel 27°C P+n (V-nb) = nRT a = 4.2 L2 atrn/mo2 b=0.037 L/mol

Using van der Waals equation, calculate the constant a when two moles of a gas confined in a four litre flask exert a pressure of 11.0 atm at a temperature of 300
Using Vander Waals equation calculate the pressure exerted by one mole of CO2. Its volume at 373 K is 0.05 dm^3 . - Sarthaks eConnect | Largest Online Education Community
Using Vander Waal's equation, calculate the constant 'a' when two moles of a gas confined in a four litre flask exerts a pressure of 11.0 - Sarthaks eConnect | Largest Online Education Community